Introduction
Ketamine is a commonly used anesthetic worldwide, in part due to its benign impact on vital functions. Ketamine is also very inexpensive relative to other anesthetic options, allowing for broader accessibility in emergency and operating rooms, even in the poorest nations.
Recent breakthroughs in the use of ketamine as an “off-label” medication for pain management and mood disorders are changing the way the medical community thinks about how this anesthetic interacts with the brain. Ketamine induces a trance-like state while also providing pain relief, sedation, and in some cases, temporary amnesia (during treatment) at high doses. At low doses, patients report feeling disconnected from their body and often come out of treatment with an elevated mood. In cases of acute suicidality, esketamine (the S(+) enantiomer of ketamine) has also been successfully administered to rapidly intervene and reduce symptoms. The variety of uses and mechanisms of action of ketamine has led to a deeper scientific understanding of the neurobiochemistry of the human brain.
Ketamine has had a complex history and reputation, from a wartime anesthetic to a notable illicit party drug, and as a result, there has been a lot of conjecture in scientific community, news, and social media over the years.
What Is Ketamine?
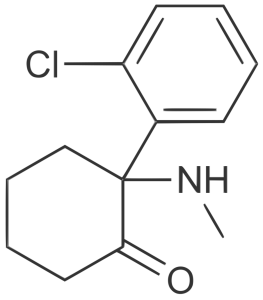 Ketamine (C13H16ClNO) is a chemical derivative of a class of compounds called arylcyclohexylamines, which have been studied and developed by pharmaceutical companies since the early 20th century for their sedative effects. Ketamine was discovered in 1956 and first synthesized in 1962 by Calvin L. Stevens. After preclinical testing in animals and humans, the Food and Drug Administration (FDA) approved ketamine in 1970 as an anesthetic for use on human beings. Armed forces medics quickly embraced it and continue to employ ketamine in combat situations today. Anecdotally, injured soldiers receiving ketamine instead of opioid medications have reported less issues with addiction and PTSD after recovery. The discovery of antidepressant effects of ketamine in 2000 has been considered an important advancement in the treatment of depression and continues to be a source of clinical study for several mood and pain disorders. In 2019, the FDA and European Commission approved a nasal spray for esketamine, under the brand name Spravato, to treat depression.
Ketamine (C13H16ClNO) is a chemical derivative of a class of compounds called arylcyclohexylamines, which have been studied and developed by pharmaceutical companies since the early 20th century for their sedative effects. Ketamine was discovered in 1956 and first synthesized in 1962 by Calvin L. Stevens. After preclinical testing in animals and humans, the Food and Drug Administration (FDA) approved ketamine in 1970 as an anesthetic for use on human beings. Armed forces medics quickly embraced it and continue to employ ketamine in combat situations today. Anecdotally, injured soldiers receiving ketamine instead of opioid medications have reported less issues with addiction and PTSD after recovery. The discovery of antidepressant effects of ketamine in 2000 has been considered an important advancement in the treatment of depression and continues to be a source of clinical study for several mood and pain disorders. In 2019, the FDA and European Commission approved a nasal spray for esketamine, under the brand name Spravato, to treat depression.
Today, ketamine is classified as a schedule III medication in the United States. The Drug Enforcement Administration (DEA) classifies substances with a low to moderate potential for psychological and physical dependence under Schedule III. These drugs can still lead to abuse or addiction when misused but are less dangerous and/or addictive than drugs in Schedules I and II. Schedule III medications are only available with a prescription. The World Health Organization (WHO) has listed ketamine on its Essential Medicines List (EML). The EML describes medications considered to be effective and safe while meeting the most important needs in a health system.
Ketamine is also classified as a dissociative anesthetic. Dissociative means a sense of disconnection from one’s ordinary reality and usual self. In other words, ketamine allows users to experience a new perspective of their usual reality. At higher dosages, ketamine can reduce pain perception and induce sedation. Ketamine can render general anesthesia at the highest of doses.
How Ketamine Works
Ketamine has been used as a medication to treat several pain and mood disorders. Although the mechanisms are not fully understood, ketamine appears to prevent neurons in the brain and spinal cord from becoming sensitized to painful stimuli. As a result, the medication has been used as both an anesthetic and an analgesic (a medication that has pain-relieving properties).
Ketamine interacts with several protein pathways in the brain important for memory, plasticity, and learning. The molecule initiates a response when combined with certain receptors, and interacts with the opioid, AMPA, GABA, cholinergic, and dopaminergic systems. As an antagonist, ketamine binds to and affects HCN1, potassium, calcium, and sodium channels in neurons, which results in the mitigation of pain responses and limits the development of chronic pain.
One of the most important mechanisms of action regarding ketamine is as an antagonist at the N-methyl D-aspartate (NMDA) receptor. Ketamine’s analgesic properties are not a result of direct action on the NMDA receptor, rather it affects a series of downstream molecular cascades. The diverse mechanisms of action may also play a role in “resetting” the pain receptors that have become hypersensitized in neuropathic pain conditions like Chronic Regional Pain Syndrome (CRPS). Because chronic pain and depression often coexist, ketamine may improve mood-related issues due to its potent antidepressant properties, which in turn, may also potentially alter pain perception in a positive way.
Traditional antidepressants work by blocking the reabsorption of brain chemicals like serotonin, norepinephrine, and dopamine to improve communication between brain cells. By contrast, ketamine increases glutamate, an excitatory neurotransmitter, when it blocks NMDA receptors. NMDA receptor antagonism leads to a downstream cascade that ultimately results in an increased production of brain proteins like Brain Derived Neurotrophic Factor (BDNF) and rapamycin (mTor). These proteins build new stronger and healthier connections, or synapses, between brain cells, reversing the damage that chronic stress and depression have on the synapses.
While more recent work has demonstrated the possibility of an antidepressant response to low doses of ketamine that produce minimal psychedelic effects, this effect tends to be more sustained with repeated use — in other words, a cumulative effect. Psychedelic, ‘dissociative’, or ‘ego-dissolving’ experiences may well be instrumental in providing a more robust effect, leading to a positive change in outlook and character that can be termed a ‘transformative’ response.
The Future of Ketamine and Mood Disorders
In 2019, the FDA and European Commission approved the use of an esketamine nasal spready under the brand name Spravato to treat depression. However, as of 2021, ketamine has not been approved by the FDA to treat depression or other mental health disorders, and therefore, the medication is being used as an “off-label” drug to treat such conditions. Off-label is a term to describe a medication that is being used beyond what it has been officially approved for by the FDA to treat conditions medically appropriate for patients. Small steps, such as the approval of Spravato, point toward a growing acceptance of ketamine as a promising tool beyond an anesthetic in the medical community.
There is a growing body of academic and clinical research regarding the mechanisms of actions (i.e. how exactly ketamine is interacting with the brain and body), safety profile (the potential risks and side effects) for prolonged repeated use, and efficacy of ketamine in treating a variety of pain and mood disorders. More information about ketamine research can be found at the ASKP3 Scientific Literature as well as the Patient Portal Key Research pages.
The promising effect that ketamine has on treating depression, specifically, will no doubt reshape the clinical treatment of mood disorders and may even provide a new framework by which novel classes of antidepressants can be developed. The mission of ASKP3 is to further awareness and education regarding safe, effective, and ethical therapeutic use of ketamine. The hope is by bringing together the medical community, the organization can create a forum by which new research can be disseminated and clinical practices can be standardized.
References:
Wei, Y, Chang, L., Hashimoto, K. (2020) A historical review of antidepressant effects of ketamine and its enantiomers. Pharmacology, Biochemistry and Behavior. 190(10):172870. DOI:10.1016/j.pbb.2020.172870
